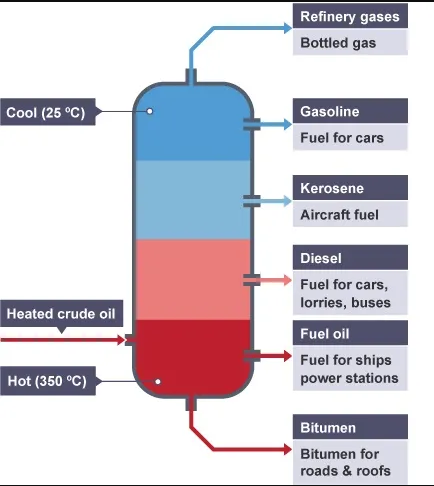
Unlike other materials with a single boiling point, such as water and turpentine, composite liquids like crude oil boil across a more comprehensive temperature range. Thousands of different compounds and elements comprise crude oil.
The initial boiling point (IBP) is the temperature at which the first distillation drop emerges, and the final boiling point (FBP), often known as the endpoint, is the temperature at which the last distillation drop appears (EP). The boiling point range for crude oil is vast, with boiling temperatures ranging from 20°C ( 68°F) to 500°C (932°F).
In this article, you will learn about the API gravity of crude oil, the various factors that affect its boiling point, and carbon number’s effect on crude oil’s boiling point.
API Gravity of Crude Oil
Like any other substance, crude oil has a collection of attributes that characterize its chemical and physical features. These characteristics allow for the comparison of different crude oil mixes. Some parts are more critical than others depending on the comparison. For example, viscosity, wax content, sulfur, and acidity are crucial in crude oil transportation and refining, whereas API Gravity is essential in crude oil trade and pricing.
The API Gravity of crude oil is a measurement of how heavy or light it is compared to water (density of crude oil/density of water). The API Gravity measurement is a positive value that is inversely proportional to the specific gravity of crude oil (the more significant the API Gravity, the lighter the crude oil).
API Gravity Classifications and Grades

Oils having an API gravity of 40–45 generally command the highest market prices. Shorter molecular chains are less acceptable to refineries in oils with API gravity of 45 or above. The four major crude oil classes are light, medium, heavy, and extra-heavy.
Light Crude Oil
Any crude oil with API gravity of over 31.1 degrees falls into the light crude oil category.
Medium Crude Oil
Oils with API gravity falling between 22.3 and 31.1 degrees are classed as medium crude oils.
Heavy Crude Oil
Heavy crude oils have an API gravity of under 22.3.
Extra-heavy Oil

Extra heavy crude oils, sometimes known as bitumen, have fewer than 10.0 degrees API gravity.
While these are appropriate classifications, it’s worth noting that the exact distinction between light, medium, heavy, and extra heavy varies depending on the origin region. Finally, fluctuations are determined mainly by the current oil commodity trade.
What affects crude oil’s boiling point?
Crude oil is a complicated hydrocarbon combination. These molecules’ carbon atoms are linked together in chains and rings. Pure hydrocarbons have different boiling points depending on their carbon number, molecular size, and hydrocarbon type (aliphatic, naphthenic, or aromatic).
Because each percentage of crude oil has its own boiling point, it boils at different temperatures and leaves the crude oil mixture as a gas when heated. There are increasing intermolecular interactions between the chains as the length of the hydrocarbon chain grows. As more energy is required to separate the chains, boiling points rise. as the size of the hydrocarbons increases.
The presence of double bonds can also affect boiling points as “trans” double bonds pack more closely together than “cis” double bonds, so they have higher melting points. At the “A” level, you have to consider the shape of the hydrocarbons, such as branches. This can reduce the boiling point compared to a straight-chain molecule.
How does crude oil carbon number affect the boiling point?
As discussed previously, crude oil is a comparatively volatile liquid hydrocarbons mixture. Regardless of differences, practically all crude oil contains between 82 and 87 percent carbon and 12 to 15 percent hydrogen.
As the number of carbon atoms increases, the boiling point also increases. This is because the force of attraction between the molecules increases as the molecule gets longer and contains more electrons.
On the other hand, chain length also affects boiling point. As the chain length (numbers of carbons) increases, these compounds’ boiling points gradually increase. Longer chain molecules have higher boiling points because longer chain molecules become wrapped around and entangled in each other much like the strands of spaghetti.



