Thermal energy refers to the energy that a body or system has due to the movement of its particles. So, is thermal energy potential or kinetic? The answer lies in examining the behavior of the moving particles of a body or system. Doing this reveals that thermal energy has both potential and kinetic forms. This article explains the potential and kinetic forms of thermal energy, how to transfer thermal energy, and its applications.
Thermal Energy as Potential and Kinetic Energy
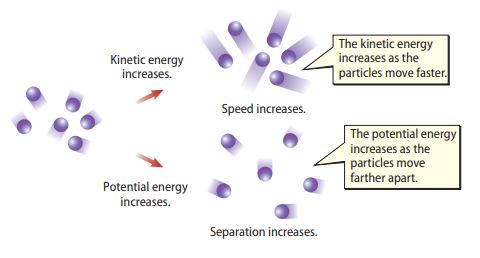
The particles of a body or system constantly vibrate and bump together. This is due to the attractive forces they exert on each other. Moreover, an increase in the body temperature drives the particles further apart. As a result, the attractive forces between them increase as well as their potential energy. In addition, the particles vibrate at greater speeds thereby increasing their kinetic energy. The combination of the potential and kinetic energy of the particles is what makes up the thermal energy of the body.
Thermal Energy and Temperature
The concept of thermal energy and temperature continue to stir up debate. From the explanation in the previous section of how potential and kinetic energy constitute thermal energy, they are clearly not the same. Nevertheless, they are closely related with the increase in one resulting in an increase in the other. In its potential form, thermal energy is a product of Boltzmann’s constant (kB) and absolute temperature (T).
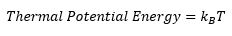
While in its kinetic form, you can quantify the change in thermal energy (Q) as a product of mass (m), change in temperature (ΔT), and specific heat (C).
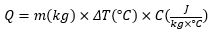
Thermal Energy Transfer
In practice, we appreciate thermal energy more in its kinetic form because it is easy to put to work. On the other hand, it is a more tedious task to do work with thermal potential energy. Heat is thermal energy in transit. It flows from an object at a higher temperature to one at a lower temperature. In nature, this occurs in three ways: conduction, convection, and radiation.
Conduction
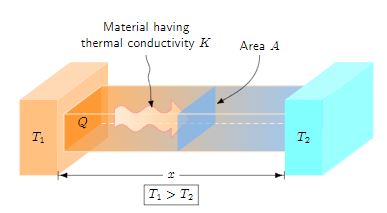
On a microscopic level, this is the transfer of thermal energy within a body as vibrating atoms and molecules interact with their neighbors. Furthermore, this energy transfer can also occur on a larger scale from one body to another. But the condition is that both bodies must be in contact. Conduction takes place in solids, liquids, and gas. However, it is most efficient in solids where molecules have a closer configuration. Fourier’s law quantifies the rate of heat transfer (Q), across a distance (Δx), as a function of the temperature difference between the bodies (ΔT), the thermal conductivity of the material (k), and the cross-sectional area perpendicular to the direction of heat flow (A).
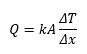
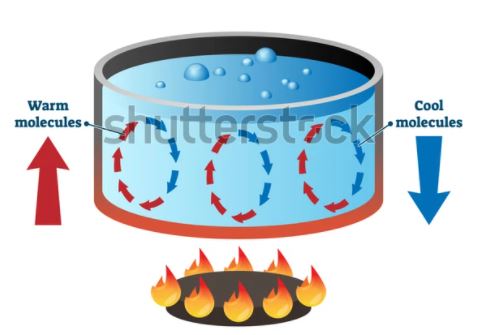
Convection
Heat transfer is classified as convection when fluid flow is the driver of the process. When a fluid receives heat, it expands and begins to flow, transferring the heat to any surface it encounters. We call this natural convection. Moreover, in applications where convection is desirable, a fan, pump or another mechanical means can increase the speed of the process. This is known as forced convection. The rate of heat transfer (Q) via convection can be quantified based on the convection coefficient of the fluid (h), contact surface area (A), and temperature difference between the fluid and the surface (ΔT).
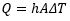
Radiation
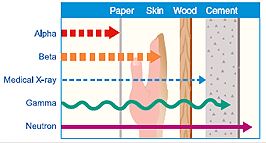
Radiation is the only means of thermal energy transfer that does not require a material medium. The heat transfer occurs by the transmission of electromagnetic waves such as alpha, beta, and gamma waves. A perfect example of radiation is that which occurs between the sun and the earth. The heat flow rate (q) from an object’s surface area (A), depends on the Stephan-Boltzmann constant (σ), emissivity (ε), the view factor between surfaces (F), and the difference of the absolute temperatures of the surfaces (Ta4 – Tb4).
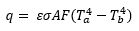
Thermal Energy Applications
In nature, several objects exhibit thermal energy in both potential and kinetic forms. The conversion of these forms of energy to do useful work are common in a variety of applications.
Solar Energy
Solar energy refers to radiant light and heat from the sun which gets to the earth through radiation. We can harness it using a range of technologies that include solar thermal energy for water heating, and solar power for electricity generation.
Solar Thermal Energy
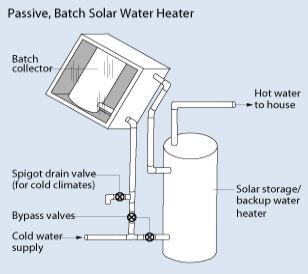
Harnessing the energy from the sun to heat up water is effective, especially for domestic use. The two ways of achieving this are through the active or passive system. In the active system, a collector which has heat exchangers absorbs heat from the sun. Then, water absorbs this heat by pumping it through the heat exchanger (direct circulation system). Otherwise, we can pump a heat-transfer fluid into the heat exchanger then use it to heat up the water located in another tank (indirect circulation system). The indirect circulation system suits temperate regions, while the direct circulation system suffices where it rarely freezes.
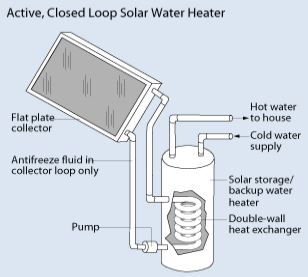
The passive system is less expensive, but also less efficient than the active system. Largely due to the absence of a pumping system. In this system, the sun heats water directly via a transparent storage tank. This hot water then flows through a plumbing system for end-use.
Geothermal Energy
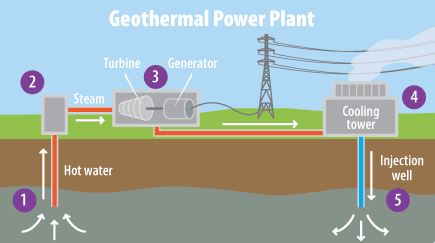
This refers to the heat energy that comes from within the earth due to the slow decay of radioactive particles in the earth’s core. As a renewable energy source, people use it for heating buildings, water, and for generating electricity.
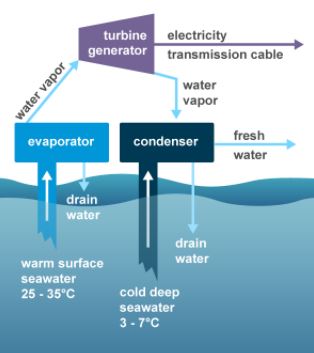
Ocean Thermal Energy
Ocean thermal energy conversion (OTEC) is one of the few processes where thermal potential energy produces useful work. OTEC technology produces energy by harnessing the temperature differences between warm ocean surface waters and cold deep ocean waters.