Thermodynamic cycles are processes that involve heat and work flowing into and out of a system. These systems usually intake a working fluid and output mechanical work or energy. These cycles use different devices to convert heat, pressurize working fluids, and burn off fuels. In this article, you will learn the principles of a thermodynamic cycle, the purpose of a cycle, and different kinds of thermodynamic cycles.
Principles Behind a Thermodynamic Cycle
Though there are many different types of thermodynamic cycles which all have different processes they share a singular goal, to be as efficient as possible. An idealized cycle has no heat loss or energy loss throughout the process, however, this is impossible to achieve. In the real world, there are inefficiencies and heat loss to the environment, and these are all accounted for in the ‘actual’ versions of these thermodynamic cycles.
In these cycles, there are also variations of the exact processes they go through. These terms help better understand what is happening in these cycles.
- Isothermal – A process which occurs at a constant temperature
- Adiabatic- A process where no matter transfer or heat transfers to or from the environment
- Isentropic- A process that is adiabatic and reversible, which produces energy as work
- Isobaric- A process that has a constant pressure
- Isochoric- A process where volume is constant
Though thermodynamic cycles differ in the exact way they manipulate heat and energy, they are all useful in different situations. Each cycle is theoretically scalable to output, no matter how much output power or work is needed. The capability of producing more energy than what enters the system is a key design factor in these systems. These systems use a working fluid to transfer heat or energy between stages. Whether the working fluid is air, water, R-134A, or even a combustible fuel, these cycles still require input energy, which is why pushing for the maximum efficiency is optimal.
Purpose of a Thermodynamic Cycle

The main purpose of a thermodynamic cycle is to extract the maximum amount of energy from the initial input of fuel. Created as a way that can capture energy from only a small amount of initial input energy. In these cycles, the compressors, pumps, and combustion chambers all require some sort of energy to run. Whether it involves combustion or electricity, they push the cycle to output more energy through the use of heat transfer. With the goal of being able to output constant energy, these cycles are designed to be repeated.
Types of Thermodynamic Cycles
Carnot Cycle
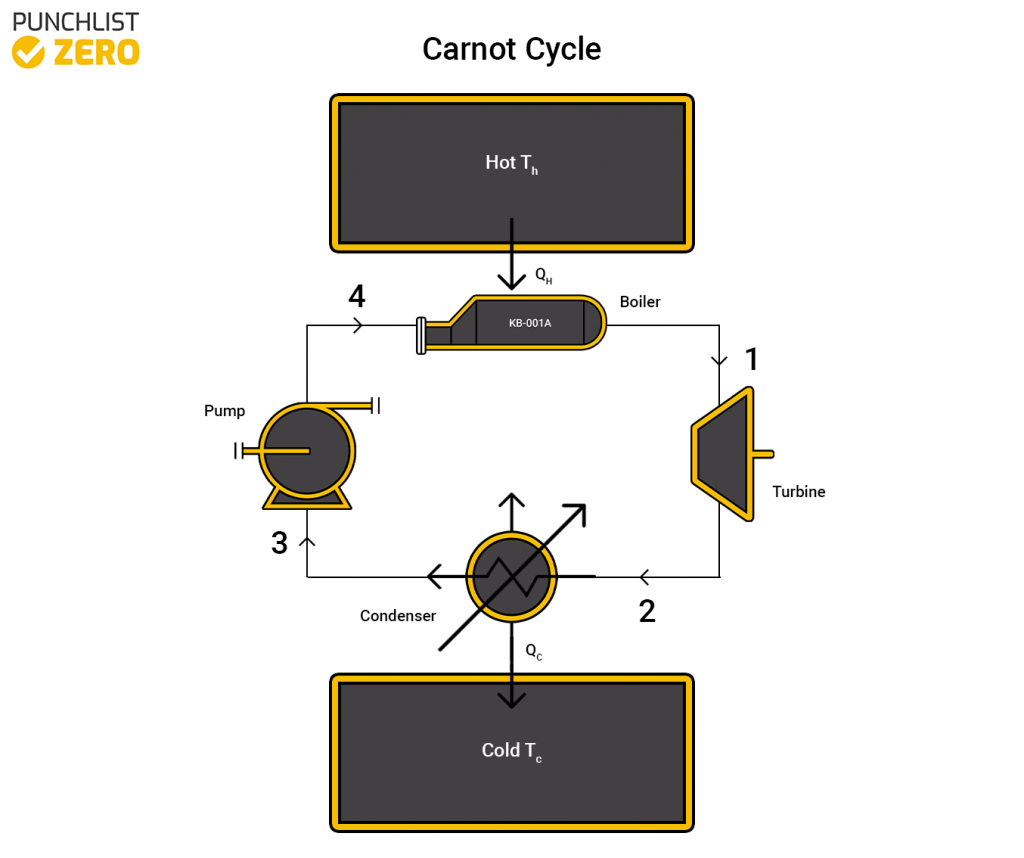
The Carnot cycle is one of the first thermodynamic cycles invented. The Carnot cycle however is only a theoretical idea because it relies on the concept of perfect efficiency. The concept of perfect efficiency is flawed because there will always be some sort of energy loss. The basic idea around the Carnot cycle is there is some sort of engine between two different heat reservoirs, a hot one and a cold one. Measuring the work done across the system is done by finding the energy difference between the Hot and Cold reservoirs.
Rankine Cycle
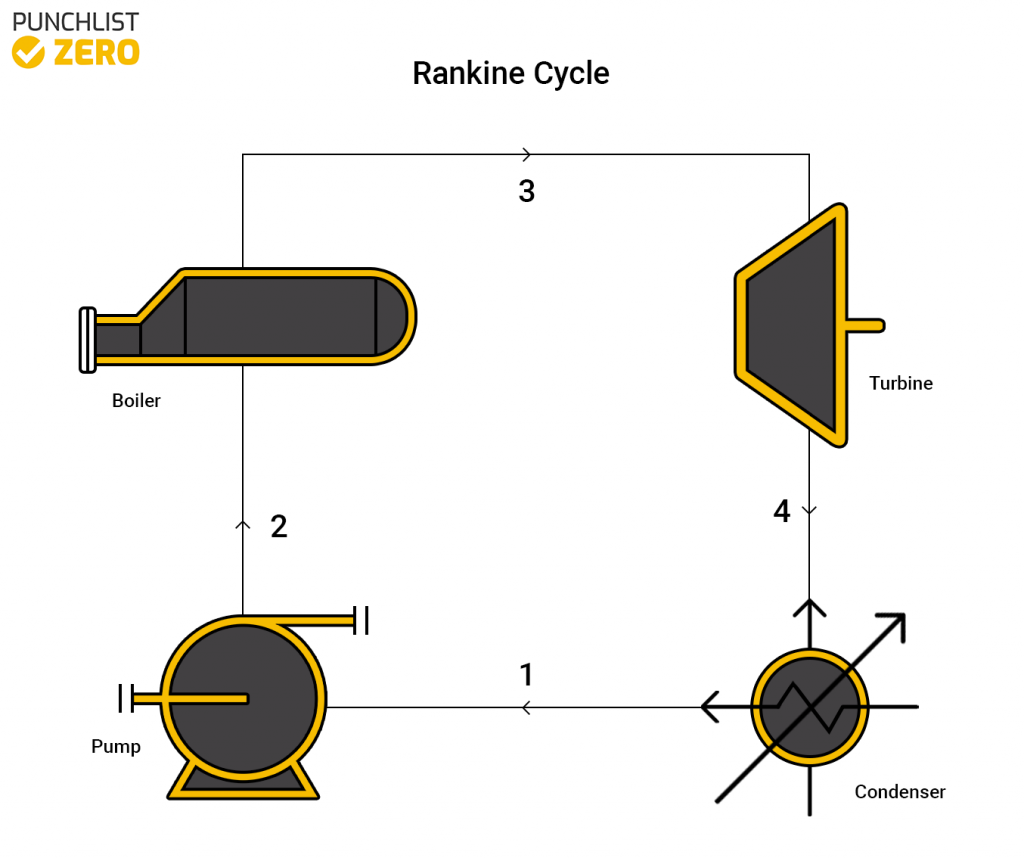
The Rankine cycle is comprised of a pump, boiler, turbine, and condenser. The first stage of the cycle involves pumping water into the system. The water heats inside a boiler until it is transformed into steam. Then the steam runs through a turbine to harvest the energy from the steam. Finally, water cools down via a condenser and returns to the pump stage. This cycle is fairly efficient however it usually involves some sort of burnable fuel to heat the water to steam effectively.
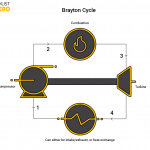
Brayton Cycle
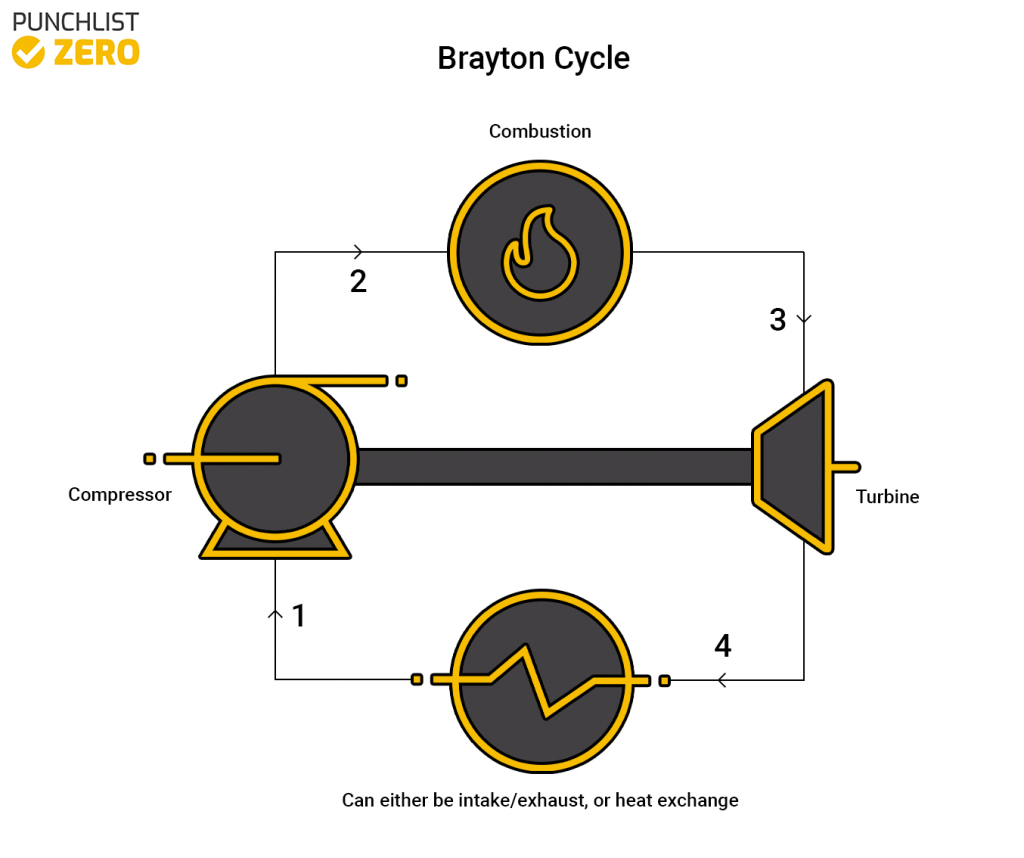
The Brayton cycle uses air as the working fluid in the system. It comprises a compressor, combustion chamber, and a turbine as the main elements. The air used can either come from fresh outside air or recycled air from the cycle. The compressor compresses the air and sends it into a combustion chamber. The combustion chamber heats the air and pushes it to the turbine. Finally, the air goes through the turbine and out of the system or back into it. Because this process doesn’t use water the only requirement to run it is some sort of fuel. This process is widely versatile and seen in many industries including the airplane industry.
Combined Cycle
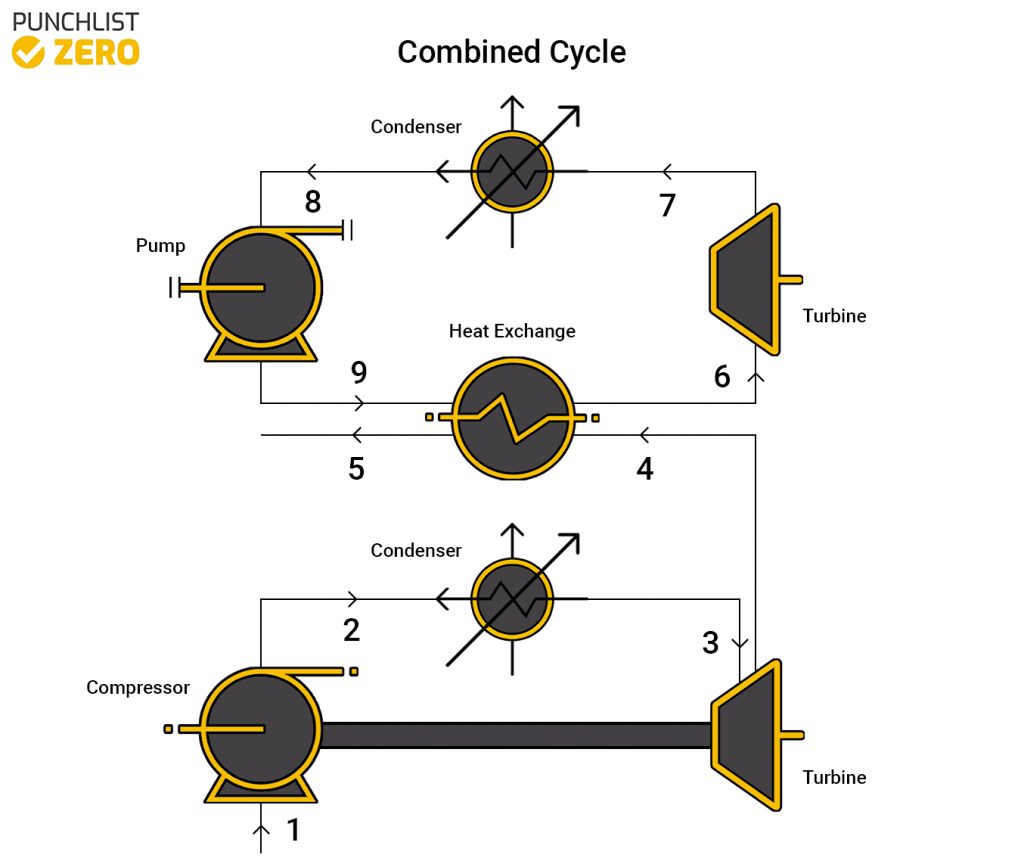
A combined cycle is the combination of the Brayton cycle and the Rankine cycle. It is used widely throughout the world on many different power plants. The cycle harvests heat from the Brayton cycle at the end of it and uses a heat exchanger to boil the water for the Rankine. Because of the heat exchange process in the system, the entire combined cycle is massively efficient, essentially only having to use fuel in one spot to power two turbines.
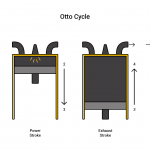
Otto Cycle
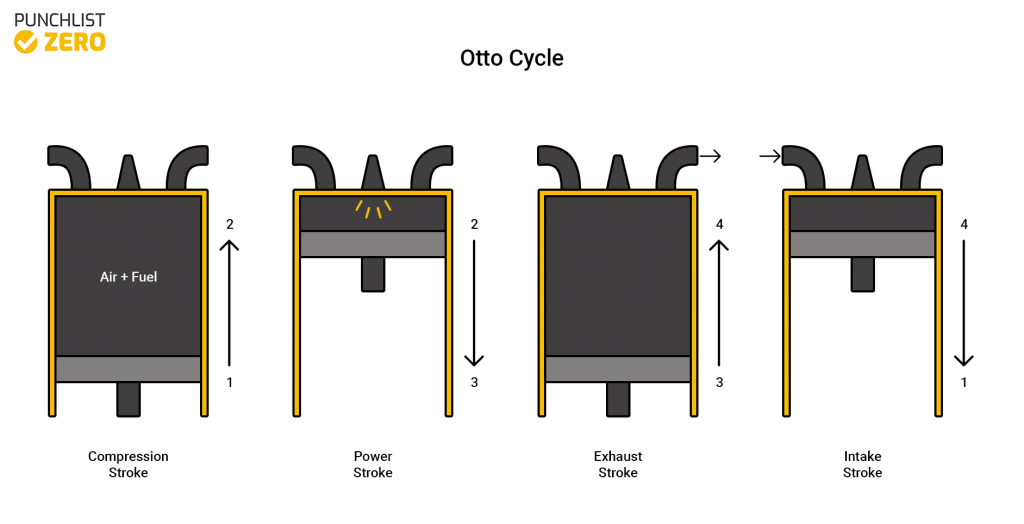
The Otto cycle is the process that shows a traditional spark ignition engine. The cycle comprises of piston-cylinder stages. The first stage, known as the intake stroke, allows the fuel and air mixture to enter the system. Next compressing the mixture in the compression stroke. Then exploding the mixture in the ignition stroke. Finally is the expansion stroke where the cylinder expands out all the way which also includes the exhaust process. Physically manifestations of the Otto cycle include internal combustion engines and reciprocating compressors.
Diesel Cycle
The diesel cycle is similar to the Otto cycle, however, it involves a higher compression ratio. The intake stroke in the Diesel cycle does not include fuel. Fuel addition occurs through a fuel injector and combusts almost immediately due to the hot air inside the cylinder. This cycle uses different fuel sources than the Otto cycle because of the higher compression ratios.
Stirling Cycle
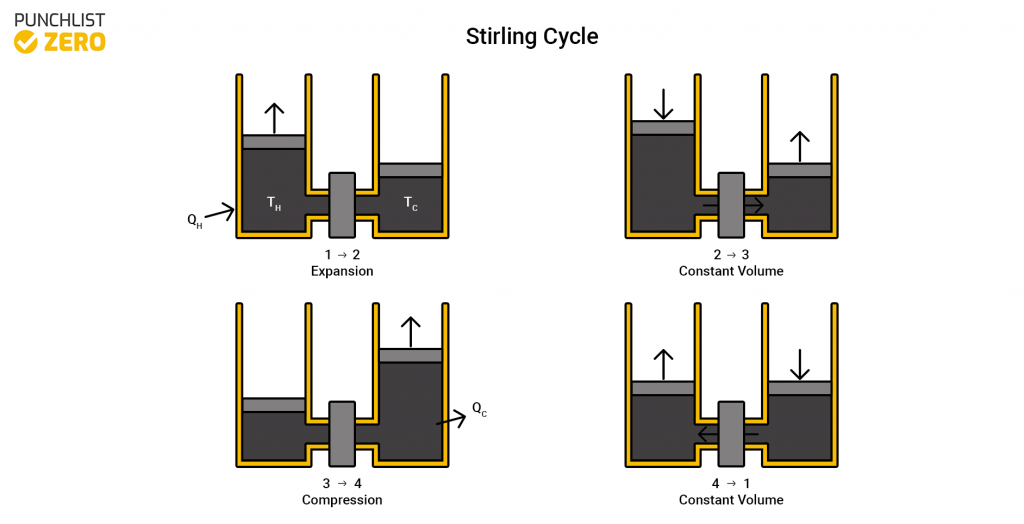
The Stirling cycle is a variation of the Carnot cycle but instead of the two isentropic processes, it contains two isochoric regeneration processes. The process is reversible which is what differentiates it from the Otto and Diesel cycles. Mechanical power may produce heating and cooling from this cycle. This cycle operates in many different stationary generators because it runs quietly and with minimal vibration.
Refrigeration Cycle
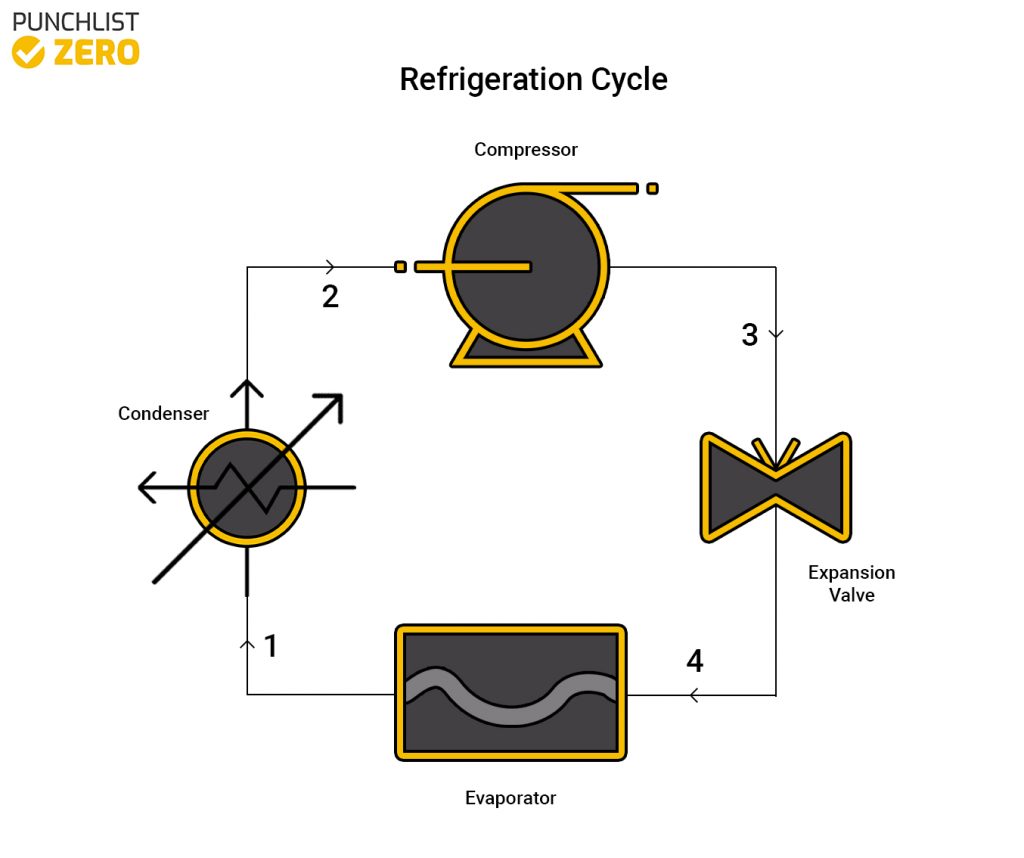
Designed to remove heat from a system rather than add it in, the refrigeration cycle keeps enclosed spaces cold. The cycle works in reverse compared to others by rejecting the heat from the system rather than using the heat. Using a compressor, condenser, and expansive valve, and evaporator, the cycle is able to effectively do this. It is traditional for the process working fluid to be a refrigerant like R134A because of its ability to remove heat effectively. Using other fluids is possible, however, many refrigerants present environmental and exposure risks.