The ductile-brittle transition temperature, often referred to as DBTT, is the temperature at which there is a pronounced decrease in a material’s ability to absorb force without fracturing. At this temperature, material fracture mode transitions from ductile to brittle. In this article, you will learn about ductility and brittleness, how ductility and brittleness influence DBTT, the transition curve, and real-world applications of DBTT.
Ductility and Brittleness
One significant change that may occur is the property of ductility. The brittle-to-ductile transition is essential for the understanding of fracture processes. Solids become brittle at very low temperatures. Their toughness becomes much higher at elevated temperatures. The transition temperature is a critical measure for the toughness of structural material.
Ductility indicates a material’s capacity to change its shape under stress without fracturing. Such plastic deformation is irreversible. This means that some fundamental structural changes have occurred during the deformation. For most materials, ductility increases with temperature.

Brittle metals do not have the capacity for plastic deformation under stress without fracturing. This means that brittle parts under extra stress can fail catastrophically without any deformation.
The distinction between brittleness and ductility is not obvious. Both ductility and brittleness depend on the material and also on their ductile-brittle transition temperature. The effect of temperature on the nature of the fracture is significant. At high temperatures, many metals show ductile fracture behavior and at low temperatures, they tend to fail by brittle fracture.
The brittle fracture is the main failure mode in the low toughness region. In this mode, there is hardly any plastic deformation before the fracture occurs. However, in the high toughness region, it is the ductile fracture where substantial plastic deformation takes place before fracture. Ductile fracture has slow propagation and it absorbs large amounts of energy before fracture.
Brittle fracture is possible only when cracks can propagate at high velocities. The crack grows along the grain boundaries and gives the fracture surface a bright crystalline or granular appearance.

Generally, the ductile fracture is the preferred failure mode. Ductile fracture provides significant plastic deformation of the object before and during the growth of cracks thus providing ample warning prior to fracture.
Ductile-Brittle Transition Temperature (DBTT)
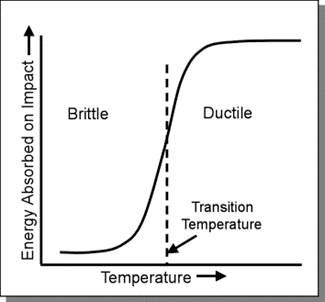
The temperature above which a material shows ductility and below which shows brittleness is the Ductile–Brittle Transition Temperature (DBTT). This temperature is not exact but depends on factors such as heat treatment during manufacturing and impurity elements. DBTT represents a point at which a metal changes from being ductile which means a high energy plastic bending or deformation state to a brittle state, which is a low energy state. It is the temperature at which there is a significant decrease in a material’s ability to absorb the stress without fracturing.
DBTT is quite evident in Body-Centered Cubic (BCC) metals, such as low carbon steel, which convert to a brittle state at low temperatures. In metals, plastic deformation occurs due to dislocation motion. The amount of stress necessary to move dislocation depends on metal crystal structures and their bonding grain boundaries. However, when overstressed, the object suffers from crack propagation and eventually fails by brittle fracture.
Ductility is an essential requirement for steel used in the construction of many steel pressure vessels. In this case, the size of the grain determines the properties of the metal. Smaller grain size increases tensile strength, increases ductility, and results in a decrease in DBTT. Proper heat management during the manufacturing of steel vessels can control the grain size. The addition of minute quantities of certain elements such as manganese and nickel to low-carbon steels also lowers its DBTT.
Ductile-Brittle Transition Curve
The ductile-brittle transition phenomenon occurs due to the occurrence of the plastic zone in some metals at a specific temperature. At high temperatures, even on application of external stress, the heat energy helps in the movement of dislocations within the crystal structure. Cracks appear at the tip in the plastic zone which then results in cracking to occur by ductile fracture.
With the decrease in temperature, the dislocation mobility and the size of the plastic zone also decrease. On reaching the transition temperature results is a big loss in fracture toughness. Below the transition temperature, the dislocation slip stops completely which results in the metal fracturing by brittle crack propagation.
The ductile-brittle transition behavior of various metals falls into three different categories determined by their yield strength and crystal structure. Metals with a Face Centered Cubic (FCC) crystal structure does not undergo the transition and retain their ductility at low temperature as they have a large number of dislocations systems in their crystal structure which lets them move, even at very low temperatures.
Aluminum is an FCC metal and, therefore, does not become brittle at low temperatures. The metals that have Hexagonal Close Packed (HCP), crystal structures such as titanium and magnesium, also do not undergo such transition. But Body-Centered Cubic (BCC) crystal structure metals often do undergo a ductile-brittle transition as fracture occurs in them brittle fracture at low temperatures and by ductile fracture at high temperature.
Examples in Real-World Situations
Gold, silver, aluminum, iron, and copper are all ductile metals that can be hammered into any shape. Non-malleable metals such as tin will break apart when struck by a hammer. Copper is quite a ductile and soft metal that may bend many times without breaking it.
Low-carbon steel containing about 0.05% to 0.30% carbon makes it malleable and ductile. It has a low tensile strength, is cheap, and rather easy to shape. Cast iron is hard, brittle, and non-malleable. It is difficult to bend or hammered into shape as it has a weak tensile strength which indicates that it will fracture easily.
At lower temperatures, the defects present in the crystal structure become less mobile and become more rigid, resulting in further reducing plasticity and ductility. For example, dipping a piece of rubber in liquid nitrogen will make it brittle, which is quite ductile otherwise at room temperature.
Ceramics show poor resistance to impact and absence of plasticity. The accidental dropping of a glass beaker or a dinner plate leads to its catastrophic damage. In ceramics, on the application of local critical threshold stress, fracture in the material takes place without prior plastic deformation.

Many major accidents throughout history have occurred as a result of brittle fractures. The most infamous of these is the catastrophic sinking of the passenger ship Titanic. The main reason the Titanic broke upon impact with the iceberg was due to the brittleness of the steel used to construct the hull of the ship. In the icy water of the Atlantic, the steel was below the ductile to the brittle transition temperature. The collision of the iceberg with the ship led to brittle fracture of the bolts that joined the steel plates of the hull. A large number of critical bolts failed, resulting in the rapid sinking of the ship.
In the aviation industry, the operating temperature range of most aircraft structures is between + 40°C and −50°C. Although the ductile-brittle transition effect is not a problem for aluminum alloys, other structural metals such as steel may be susceptible. Any aircraft material should not turn brittle within these temperature ranges otherwise the aircraft structure may be susceptible to damage. DBTT is one of the important thermal properties to consider in material selection. This crucial property provides failure mechanism insight into manufactured objects and their related causes such as overload, over-stress, fatigue, or harsh environmental conditions. This is extremely important for critical parts manufactured for structural applications, especially in low‐temperature conditions.